PREMIUM IMMUNE CELLS FOR CELL & GENE THERAPY RESEARCH AND DEVELOPMENT
GETTING CELL & GENE THERAPIES TO MARKET FASTER WITH CUSTOM SOLUTIONS
Immune Cell Products:
The field of cell & gene therapy continues to hold remarkable promise for the treatment of cancer and other chronic conditions. With more than 40 years of biospecimen collection, BioIVT is ready to be your preferred provider for immune cell products. Whether you’re developing a cell or gene therapy or the manufacturing platforms to support it – we’ll work with you to ensure you get the cells you need when you need them.
THE RIGHT STARTING MATERIAL FOR YOUR CELL AND GENE THERAPY RESEARCH
With cell & gene therapies, the process is the product – so ensuring high-quality starting material is a critical first step in maximizing your results. BioIVT has over 40 years of experience in biospecimen procurement to bring you the best products for your research. Our diverse donor pool consists of over 600 recallable, consented donors around the globe. All products are collected using strict IRB protocols at strategically located facilities under FDA and HTA guidelines.
Choose from any of the following immune cell products:
RUO & GMP Leukopaks
LEUKOMAX® and VivoSTART™ leukopaks from both normal and disease state donors
View Options
For our collection process and cancellation policies, see our Leukapheresis Collections Page.
Mononuclear
Cells
Mononuclear Cells, from Peripheral Blood (PBMC), Bone Marrow (BMMC) and Cord Blood (CBMC)
View Inventory
Purified Immune Cell Subsets
Purified Immune Cell Subsets, isolated from Peripheral Blood, Bone Marrow and Cord Blood
View Inventory
Look to BioIVT for expertise in the processes required for the ethical acquisition of biospecimens
and the protocols needed for the production of reliable data.
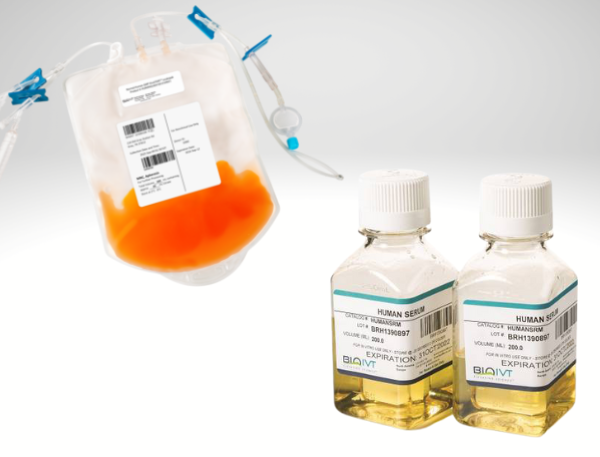
GMP MANUFACTURING WITH VIVOSTART™
With cell & gene therapies, the process is the product – so ensuring high-quality starting material is a critical first step in maximizing your results. BioIVT’s new line of VivoSTART GMP-compliant leukopaks along with plasma-derived/off-the-clot human AB serum can seamlessly integrate into your workflow.
- Collections performed in an FDA-registered, GMP-compliant center that meets 21 CFR 1271 specifications
- Batch record documentation in compliance with FDA and HTA requirements
Maximize your GMP immune cell expansion with VivoSTART Human AB Serum to reach therapeutic doses faster.
.png?width=960&height=714&name=IC_Email%20Infogaphic%20(1).png)
THE RIGHT MATERIAL FOR YOUR CELL AND GENE THERAPY RESEARCH
Choose from any of the following immune cell products:
- LEUKOMAX & VivoSTART Leukopaks
- Normal, Mobilized, and Disease-State
- Purified Immune Cell Subsets
- Isolated from peripheral blood, bone marrow, and cord blood
- Human AB Serum
Look to BioIVT for expertise in the processes required for the ethical acquisition of biospecimens and the protocols needed for the production of reliable data.

GET CLOSER TO REAL-WORLD CONDITIONS WITH DISEASE STATE IMMUNE CELLS
Choose from any of the following disease states:
- Oncology – including ALL, CLL, DLBCL, MM, solid tumors, and more
- PBMCs and limited LEUKOMAX Leukopaks available
- Autoimmune/Inflammatory – including SLE, RA, Crohn’s, and more
- PBMCs and Half/Quarter LEUKOMAX Leukopaks available
- Infectious Disease – including HBV, HCV, HIV, and more
- PBMCs and Half/Quarter LEUKOMAX Leukopaks available

HIGH-DEFINITION CHARACTERIZATION TO GET THE MOST OUT OF YOUR CELLS
Our standard high-resolution characterization capabilities include:
- HLA-typing – 6-digit typing on HLA-A-c, DPA & DPB, DQA & DQB, and DRB
- Fc-receptor genotyping – integral part of the antibody-dependent cellular cytotoxicity (ADCC) pathway
- CMV serology status – a common opportunistic pathogen, cytomegalovirus infection can impact subsequent immune responses
Get the product information you need for your research with our technical content. Click here to learn more about our solutions for cell and gene therapy research with our suite of white papers,
videos, blogs and more.

[WEBINAR] Critical Considerations When Sourcing Cellular Starting Materials & Ancillary Products for CGT

[WHITE PAPER] Disease State Primary Cells Bring Verification and Validation Studies Closer to Real-World Results

[VIDEO] Cell & Gene Therapy Insights Fastfacts: Getting Closer to The In Vivo Environment With Human Ab Serum

[PROTOCOL] Thawing Cryopreserved LEUKOMAX Leukopaks

[BROCHURE] Immunology & Immune Cell Products
Download our brochure to learn more about our portfolio of immune cell products has your cell & gene therapy, regenerative medicine, or immunotherapy research needs covered.

[COMPENDIUM] Selected Immune Cell Publications
BioIVT's Immune Cell Subsets and Leukopaks have incredible utility in many different research applications. See how they've helped researchers like you!
